
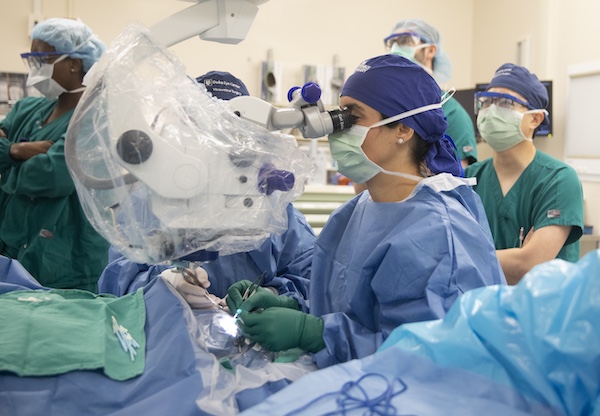
Duke Eye Center has achieved a historic milestone as the first U.S. academic medical center to implant a newly FDA‑approved, cell‑based gene therapy for macular telangiectasia type 2 (MacTel), a rare, progressive retinal disease that gradually erodes central vision.
For decades, people diagnosed with MacTel faced inevitable vision loss with no treatment capable of slowing progression. That changed in March 2025, when the FDA approved revakinagene taroretcel‑lwey (Encelto™), the first therapy proven to preserve photoreceptors, the light‑sensing cells essential for vision. The encapsulated cell implant releases protective proteins into the retina, aiming to slow degeneration and allow patients to maintain their remaining sight.

“This implant offers real hope for patients facing vision loss because of MacTel,” said Lejla Vajzovic, MD, professor of ophthalmology, who led the surgical team. “We are helping patients live fuller, more independent lives by preserving their vision.”
The implant is approximately the size of a grain of rice and houses more than 200,000 living cells within a permeable membrane. While it cannot reverse existing damage, it represents a major step forward in stabilizing disease progression.
The first recipient at Duke, Yvette Crawley said, “This disease is relentless. Because of the research that led to this treatment, I can continue to make choices in my retirement life without being hindered by vision loss.”
Duke Research Leads to FDA‑Recognized Endpoint for Retinal Trials
The approval of revakinagene taroretcel‑lwey was made possible in part by Duke’s innovative research and leadership in the clinical trials. The Duke Reading Center, led by Glenn Jaffe, MD, Robert Machemer M.D. Distinguished Professor of Ophthalmology and vitreoretinal disease division chief, in collaboration with Eleonora Lad, MD, PhD, and Sina Farsiu, PhD, determined loss over time in the ellipsoid zone on optical coherence tomography (OCT), a new structural endpoint, that became central to the FDA approval.
MacTel, once characterized primarily as a rare vascular condition affecting roughly 0.1% of the population, is now understood as a more common neurodegenerative disease involving photoreceptors. Research at Duke was instrumental in reframing the understanding of disease progression on imaging.
“A breakthrough moment came with the focus on the Ellipsoid Zone (EZ), a mitochondria‑rich photoreceptor layer visible on OCT, EZ loss signals photoreceptor damage associated with measurable vision loss, making it an ideal biomarker of disease progression,” said Lad, professor of ophthalmology and vice chair of clinical research.
Duke researchers demonstrated that the rate of EZ loss on OCT correlates nearly 100% with functional vision decline, establishing a robust structural marker of disease activity. This evidence helped the FDA formally recognize EZ loss as a surrogate endpoint for retinal disease trials, including MacTel and geographic atrophy. This shift marks a new era in retinal therapeutics — one that relies on structural biomarkers to accelerate drug development and approval.
“This milestone reflects years of rigorous research and collaboration,” said Jaffe. “We’ve not only helped bring the first treatment for MacTel to patients but also opened new pathways for evaluating therapies in other retinal diseases.”

Advancing AI to Power Future Retina Trials
Duke researchers also drove advances in artificial intelligence to support endpoint development and clinical trial analysis. Led by Sina Farsiu, PhD, Anderson-Rupp Professor of Biomedical Engineering at Pratt School of Engineering and Professor of Ophthalmology, four of his students and postdocs developed software and AI technologies for analyzing the clinical trial data. Stephanie Chiu, PhD, a former student, and Dibyendu Mukherjee, PhD, a former postdoc, developed the software that was used in this trial to quantify EZ loss on OCT and correlate it with functional vision decline.
Further, Jessica Loo, PhD, a former student, created an AI system that accurately assessed real‑world clinical trial data, performing on par with expert human graders, and Zane Zemborain, a current PhD student, developed AI‑enabled tools to identify key retinal biomarkers with unprecedented efficiency. These methods have been published in top scientific journals, demonstrating that AI can replicate clinical trial outcomes with high consistency.

The work ran in parallel with the MacTel study and has implications for a wide spectrum of retinal diseases, including retinitis pigmentosa and geographic atrophy.
From Concept to Clinic
With the first academic medical center implant now completed, after decades, the science has come full circle, from discovery, to clinical trial leadership, to FDA approval, to patient care.
“This is Duke’s mission in action — turning research into real treatments,” Lad said.